A new nuclear medicine therapy can cure human non-Hodgkin lymphoma in an animal model, according to research published in the April issue of The Journal of Nuclear Medicine. A single dose of the radioimmunotherapy, [177Lu]Lu-ofatumumab, was found to quickly eliminate tumor cells and extend the life of mice injected with cancerous cells for more than 221 days (the trial endpoint), compared to fewer than 60 days for other treatments and just 19 days in untreated control mice.
Non-Hodgkin lymphoma is a common blood malignancy. The American Cancer Society estimates that more than 80,500 new cases and 20,100 deaths will occur in the United States in 2023. The standard of care for many non-Hodgkin lymphoma patients involves chemotherapy and immunotherapy targeting the CD20 protein, which is highly expressed on most non-Hodgkin lymphoma cells.
“Although this chemotherapy with immunotherapy combination is usually initially effective, many patients don’t respond or relapse, so we need improved therapies,” said Richard L. Wahl, MD, the Elizabeth E. Mallinckrodt Professor and director of Mallinckrodt Institute of Radiology at Washington University School of Medicine in St. Louis, Missouri.
In the study researchers labeled ofatumumab, a recently developed anti-CD20 fully human antibody, with 177Lu, a widely used therapeutic radioisotope that can kill cancer cells. They then determined the in vitro characteristics of [177Lu]Lu-ofatumumab, estimated human dosimetry, and evaluated its therapeutic effectiveness for non-Hodgkin lymphoma in a mouse model.
The most significant finding of the study came from the preclinical therapy trial. Mice injected with human B cell lymphoma were either untreated, treated with unlabeled ofatumumab, treated with 8.51 MBq of [177Lu]Lu-IgG, or treated with 0.74 MBq or 8.51 MBq of [177Lu]Lu-ofatumumab. The best results occurred with 8.51 MBq of [177Lu]Lu-ofatumumab, for which the median survival was more than 221 days, essentially curing the mice. The median survival of untreated mice and those treated with unlabeled ofatumumab, [177Lu]Lu-IgG, and 0.74 MBq of [177Lu]Lu-ofatumumab was 19, 46, 25 and 59 days, respectively.
“What’s more, in mice treated with 8.51 MBq of [177Lu]Lu-ofatumumab, detectable tumors were eliminated completely within two days. Mice treated with the other therapies or left untreated, on the other hand, continued to show tumor cells present,” Wahl explained.
Researchers were able to produce [177Lu]Lu-ofatumumab with high yield and high purity. It showed favorable in vitro characteristics and dosimetry estimates that support the feasibility of human translation.
“The excellent therapeutic results in this animal model of human B cell lymphoma suggest that this curative treatment should be tested in humans with non-Hodgkin lymphoma,” noted Wahl. “If testing is successful in humans, this would represent an excellent new treatment option for patients with this disease.”
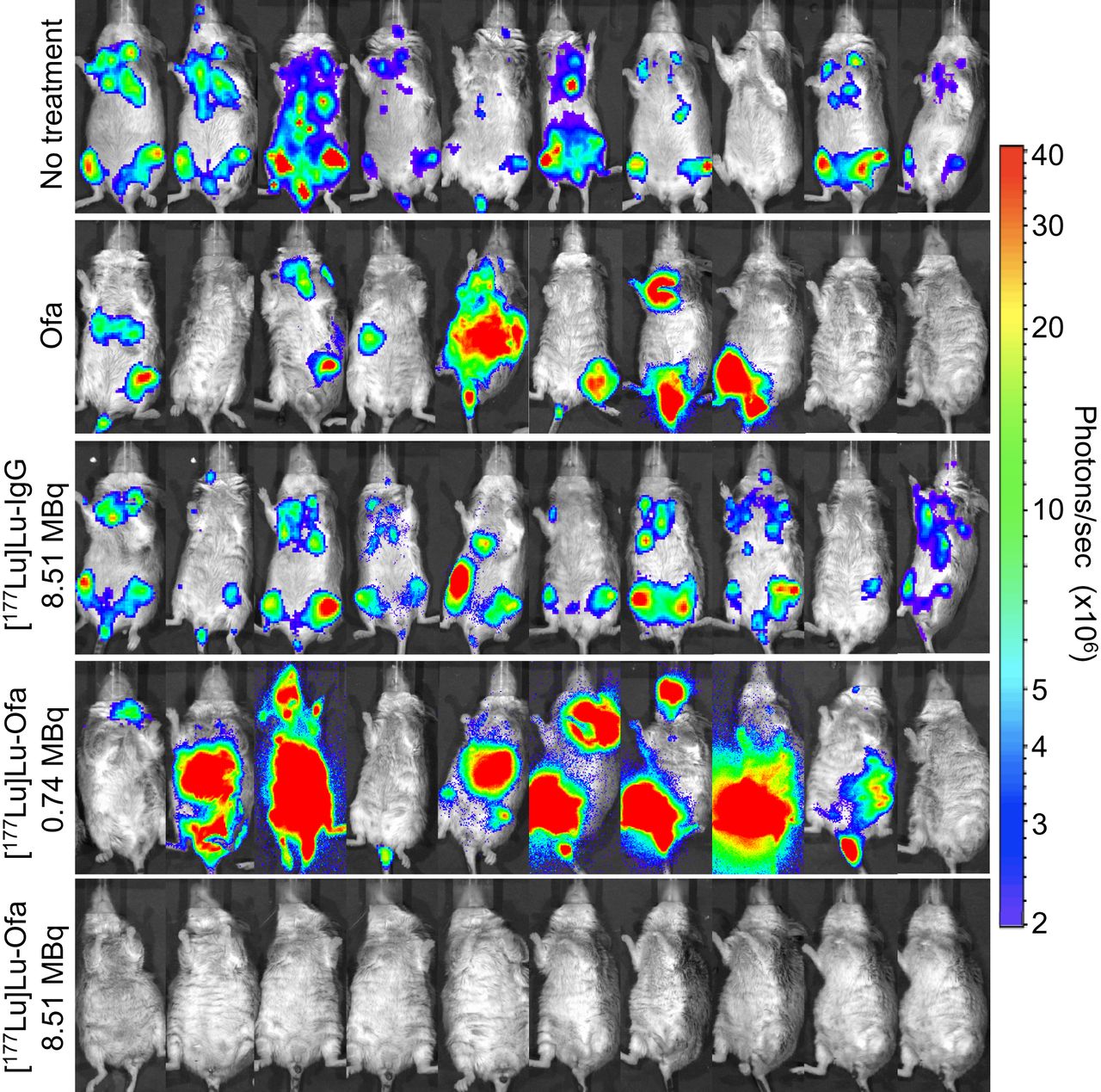
Figure 4. Bioluminescence images of untreated and treated mice with human B cell lymphoma. Optimal results occurred with 8.51 MBq of [177Lu]Lu-ofatumumab (bottom row). Ofa = ofatumumab.